remergs.com
SELECTIVE SEROTONIN RE-UPTAKE INHIBITORS GENERAL INFORMATION Antidepressant of choice for new prescriptionsSafer in overdose :. much more popular than TCAsUses: depression, OCD, panic disorder, social anxiety, PTSD, Bulemia, social phobia, migrainesDrug interactions are very common and important: most are potent P450 inhibitorsTremors, headaches, nervousness, anxiety, insomnia, an
 NATIONAL
NATIONAL QUESTION 3 (Start on a new page.)
QUESTION 3 (Start on a new page.)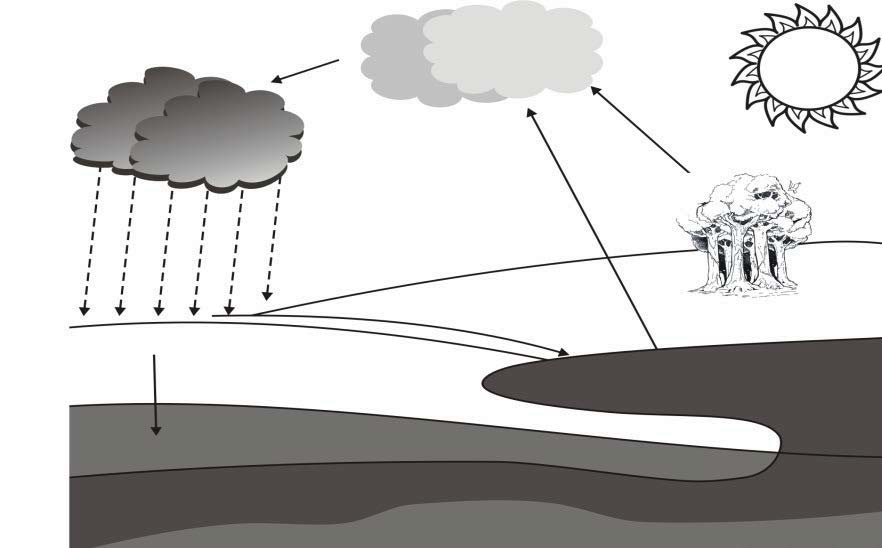 QUESTION 9 (Start on a new page.)
QUESTION 9 (Start on a new page.)